The adsorption principle of molecular sieve is based on the size and shape of the pores inside the crystals. The size of its internal pores determines its ability to adsorb and repel molecules of different sizes and polarities.
Besides molecular sieve, activated carbon, silica gel, and activated alumina are also commonly used adsorbents. In order to better understand molecular sieves, it is necessary to know some basic knowledge of adsorption, desorption and adsorbents.
Adsorbents, Adsorbates and Adsorption
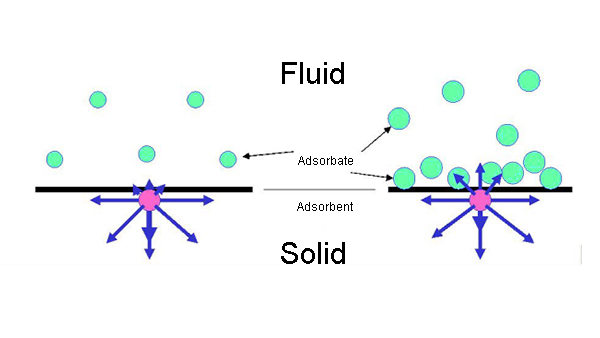
When a fluid (qas or liquid) comes into contact with a solid porous substance, one or more components in the fluid are transferred to the outer surface of the porous substance and the inner surface of the micropores, and are enriched on these surfaces to form amonomolecular layer or a multi-molecular layer. This process is called adsorption.
The adsorbed fluid substance are adsorbates, and the porous solid substance are adsorbents.
Adsorption and Separation
Due to the different physical and chemical properties of the adsorbents and adsorbates, the adsorbents’ adsorption capacities for different adsorbates, are also different.
When a fluid comes into contact with an adsorbent, the adsorbent has a higher adsorption selectivity for one or some components in the fluid, relative to other components. The components of the adsorbed phase and the residual phase can be enriched, thereby realizing the separation of substances.
Adsorption and Desorption
Adsorption can be considered as a concentration process or a liquefaction process. Therefore, the lower the temperature and the higher the pressure, the greater the adsorption capacity. For all adsorbents, gases that are more easily liquefied (higher boiling points) have a greater adsorption capacity, and gases that are less likely to liquefy (lower boiling points) have a lower adsorption capacity.
Desorption can be considered as a process of gasification or volatilization. Therefore, the higher the temperature and the lower the pressure, the more complete the desorption will be. For all adsorbents, gases that are more liquefied (higher boiling points) are less likely to desorb, and gases that are less liquefied (lower boiling points) are more likely to desorb.
Physical Adsorption and Chemical Adsorption
Principle of physical adsorption separation: Separation is achieved by utilizing the difference in adsorption force (van der Waals force, electrostatic force) between atoms or groups on the solid surface and foreign molecules. The size of the adsorption force is related to the properties of both the adsorbent and the adsorbate.
Principle of chemical adsorption separation: lt is based on the adsorption process in which a chemical reaction occurs on the surface of the solid adsorbent to combine the adsorbate and the adsorbent with chemical bonding force, so it has strong selectivity. Chemical adsorption is generally slow, can only form a monolayer and is irreversible.
For example:
- Molecular sieves rely on the special pore structure and the Coulomb force field between the equilibrium cations and the molecular sieve skeleton to produce adsorption. They have good thermal and hydrothermal stability and are widely used in the separation and purification of various gas and liquid phases. The molecular sieve adsorbents have the characteristics of strong selectivity, high adsorption depth and large adsorption capacity.
- Activated carbon relies on the van der Waals force generated with the adsorbate to produce adsorption. It is mainly used for adsorption of organic compounds, adsorption and removal of heavy hydrocarbon organic matter, deodorants, etc.
- Silica gel relies on the abundant surface hydroxyl groups inside the pores to produce chemical adsorption, and also relies on the van der Waals force between the silica gel surface and the adsorbate to produce physical adsorption.
- Activated alumina is mainly physical adsorption. In the activation process, hydrated activated alumina will quickly dehydrate, forming a form with highly developed pore structure and specific surface area, and has strong adsorption capacity for water. Both the adsorption and desorption rates of activated alumina are very fast.






